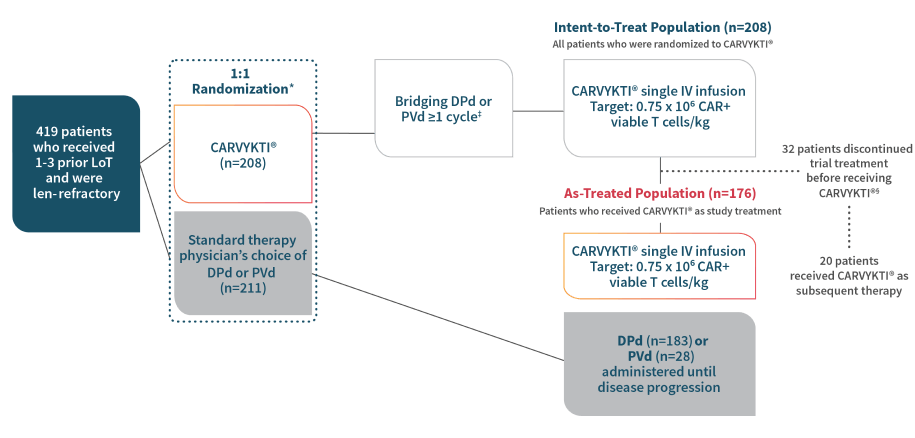
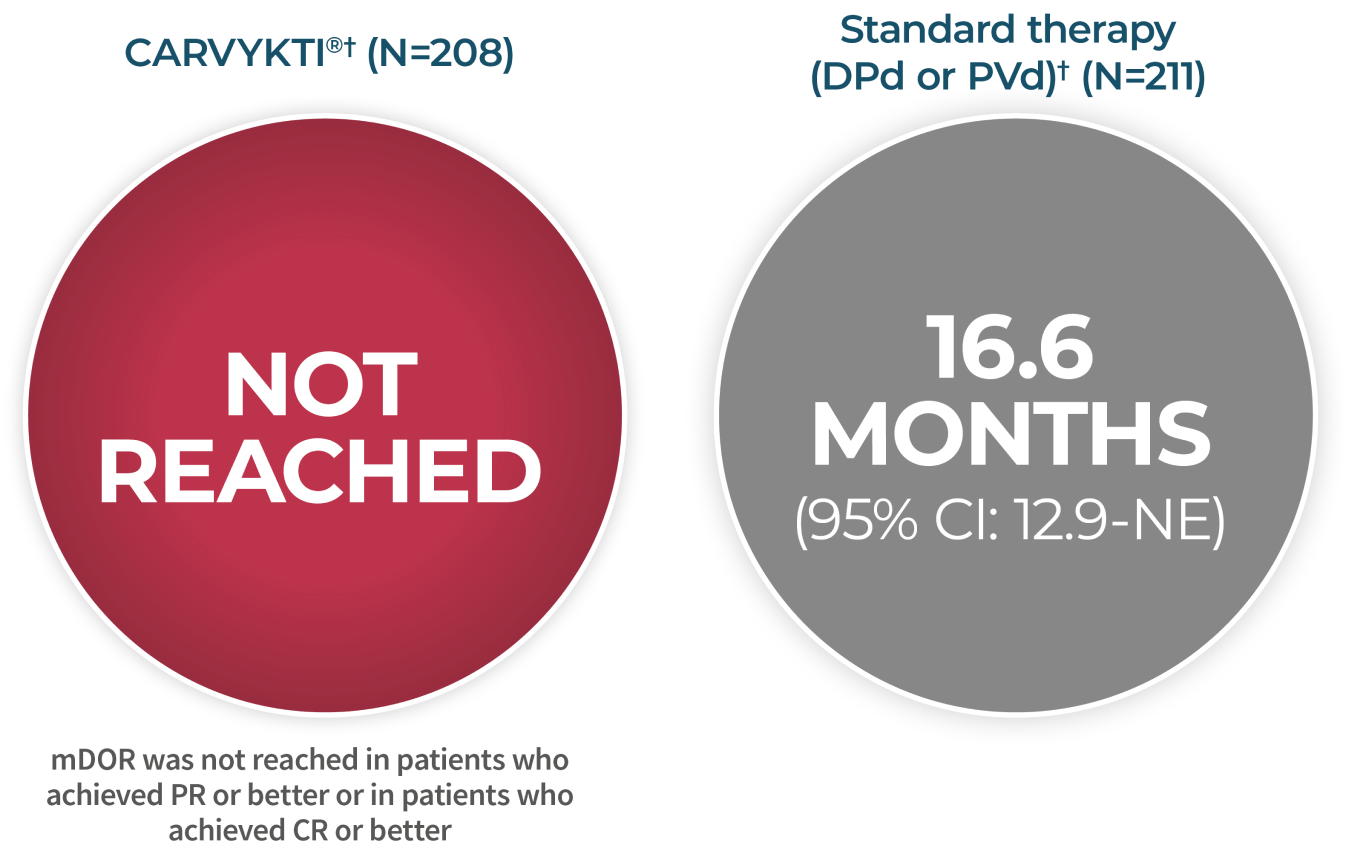
Powerful results
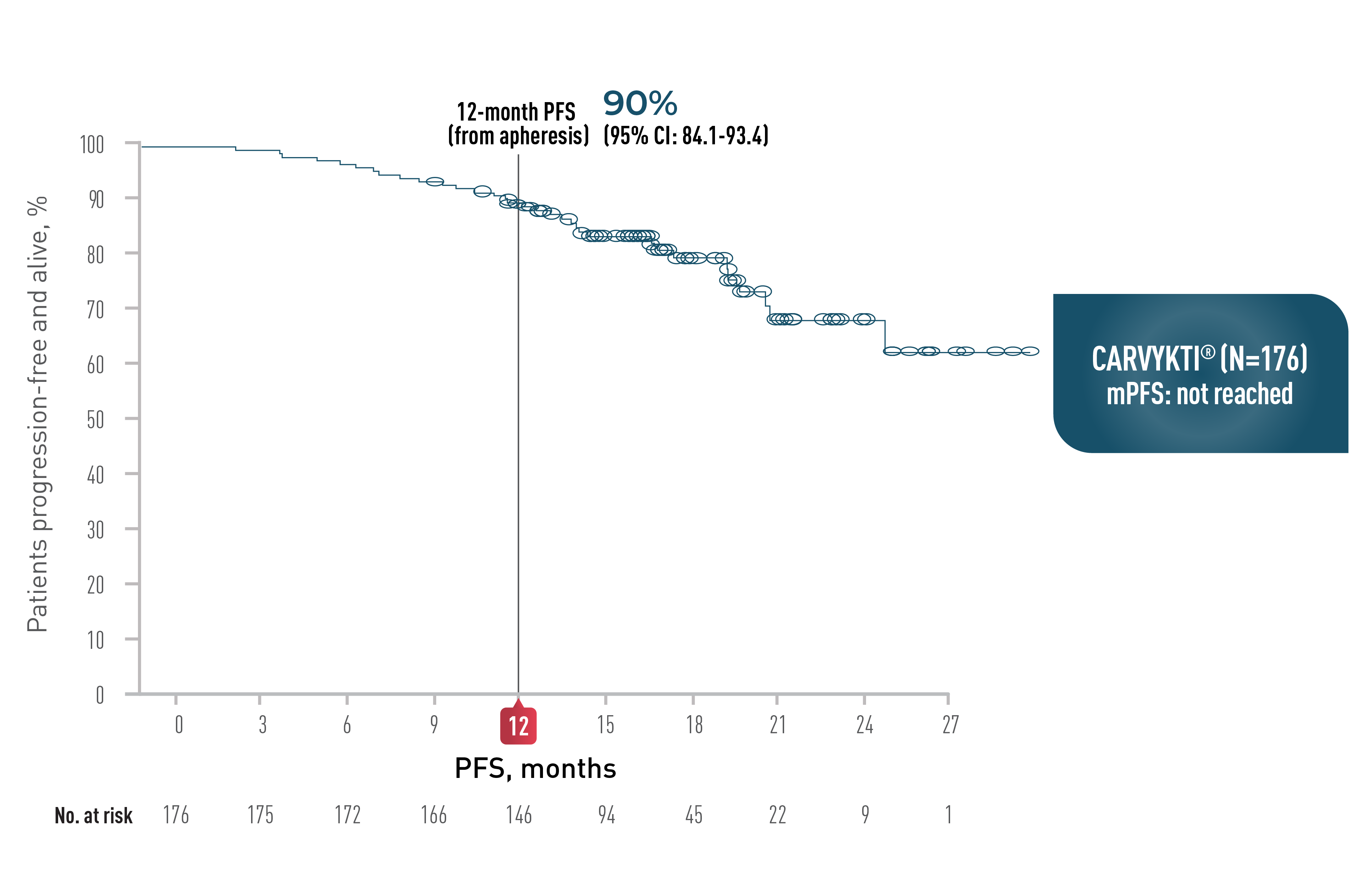
CARTITUDE-4 PFS at 15.9 months
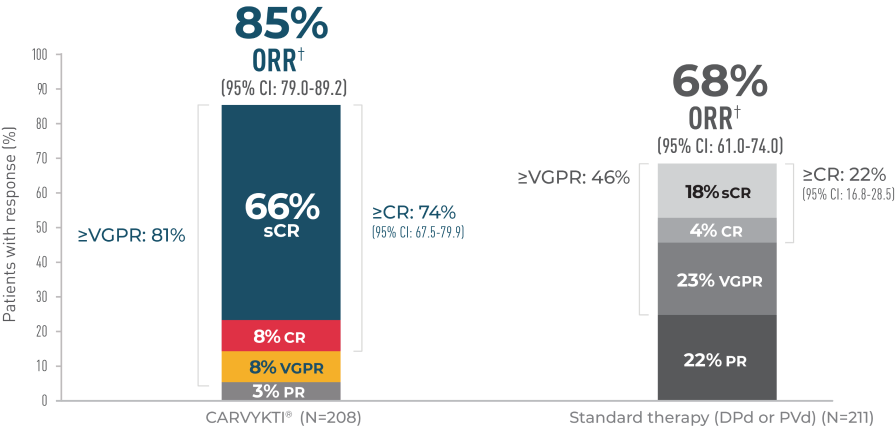
CARVYKTI® significantly prolonged progression-free survival (primary endpoint) vs standard therapy (DPd or PVd)1-3*
Progression-free survival
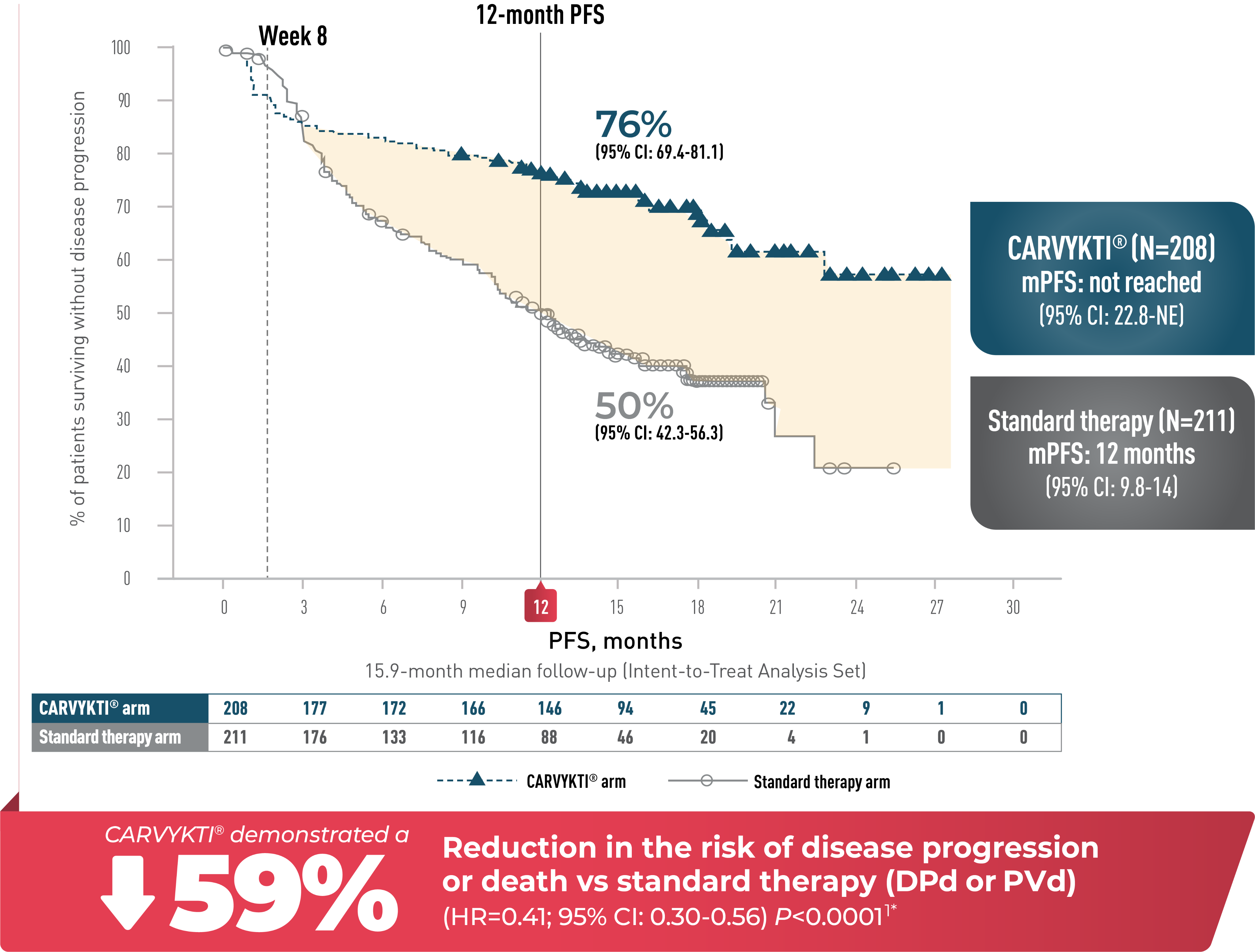
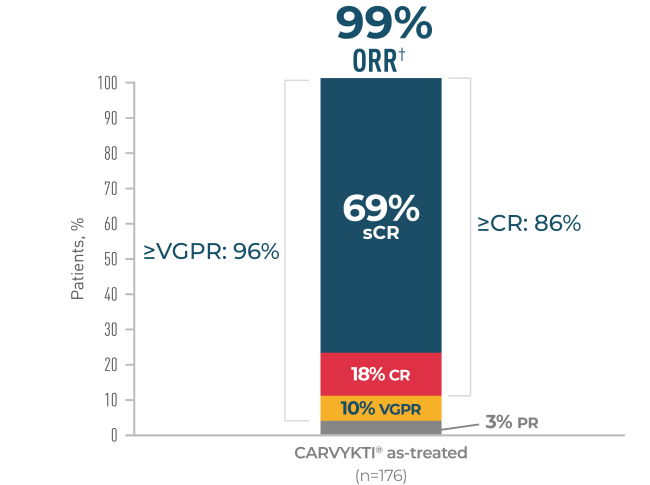
Percentages rounded to nearest whole number.
CI=confidence interval; DPd=daratumumab, pomalidomide, and dexamethasone; HR=hazard ratio; mPFS=median progression-free survival; NE=not estimable; PFS=progression-free survival; PVd=pomalidomide, bortezomib, and dexamethasone.
*Median follow-up was 15.9 months in the Intent-to-Treat Analysis Set.